|
3/23/2023 0 Comments Entropy increases 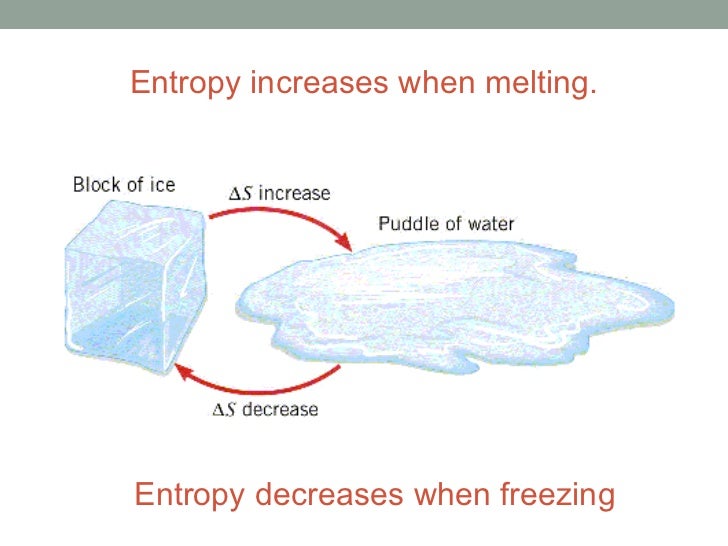
This system is realized clearly for a binary mixture of hard spheres in a box with reflective walls. While particle-particle and particle-wall attraction energies affect the distribution of particles, we show that the emergent entropic forces - quasi-gravitational - have a significant contribution to the configuration of the system. Then we employ molecular dynamics simulations to extract the effect of entropy and energy on the binary mixture distribution and how they compete with each other to determine the system's configuration. We intuitively show that this is a consequence of maximizing the accessible states for fluctuating degrees of freedom as a portion of excluded volumes reside outside of the box when they locate near the wall. First, using a model, we intuitively show that large particles prefer to position at contact with the wall as it accompanies an increase of the system's entropy.  
Here, we study how the energy and entropy affect the distribution of a two-component system in a box. Therefore, infinite knowledge of these two quantities leads to precise prediction of the system's trajectories. Why can the world resist the law of entropy increase and produce self-organizing structure Does the entropy of an isolated system always only. The energy and entropy, expressed in free energy, determine the behavior of a system. The relationship between the Helmholtz free energy F, total energy U and waste heat TS ( T : temperature, S : entropy) can be written. The second law of thermodynamics (entropy increase) is represented by the diagonal lines of the pyramid – the boundary between useful free energy and waste heat. This constant energy is represented by the constant width of (Figs. Since there are no net fl ows of energy between large (>100 Mpc 3 ) comoving volumes of the universe, energy is conserved ( fi rst law of thermodynamics). This is how entropy increases (Jaynes, 1984 ). Energy is conserved, but distributing it over a larger number of degrees of freedom makes it less extractable to do work. In this way, free energy is converted into waste heat by dissipative structures, and the overall ability to do useful work diminishes. Just as big atmospheric whirls on Earth dissipate into little whirls and soon become microscopic waste heat, on a cosmic scale, the energy of the universe – initially stored in a small number of degrees of freedom – dissipates as it spreads out over a larger number of degrees of freedom (Fig. Figure 2 is a more explicit version of Fig. While Earth-bound climate scientists take the free energy from the Sun as a given, astrophysicists can dig deeper into the origin of free energy. conversion of free energy into waste heat can be similarly described for all processes (Kleidon, 2010 ). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
